Worldwide
Login- Research
- 1x1 Platforms
- 1x1 Overview
- tDCS Overview
- 1x1 tDCS Device
- 1x1 tES Device
- 1x1 Clinical Trials Device
- 1x1 Accessories
- High Definition Platforms
- HD Overview
- HD-tDCS Overview
- 4x1 HD Adaptor
- MxN-5 REMOTE
- MxN-PRO
- MxN-GO EEG
- HD-Accessories
- Releasenotes MxN-PRO
- Releasenotes MxN-GO EEG
- REMOTE
- REMOTE Overview
- mini-CT
- SNAP Headgear
- ElectraRx
- MOBILE
- Releasenotes ElectraRx
- Software
- Neurotargeting Overview
- HD-Explore
- HD-Targets
- HD-Targets-IFS
- tDCS-Explore
- Modeling Service
- Releasenotes HD-Explore
- Releasenotes HD-Targets
- Transcranial Focused Ultrasound Stimulation (tFUS)
- tFUS Overview
- Quality Assurance Tester
- Transcranial Pulse Stimulation (TPS)
- Temporal Interference Stimulation (TI)
- Temporal Interference Stimulation (TI)
- Releasenotes TI
- HIGH DEFINITION ECT
- TRANSCRANIAL PHOTOBIOMODULATION
- transcutaneous spinal DCS (tSDCS)
- Multimodal Solutions
- Multimodal Overview
- tES + PET
- tES + fMRI
- tES + fNIRS
- tES + EEG
- fNIRS + EEG
- Galvanic Vestibular
- GVS Devices
- GVS Accessories
- Transcranial Magnetic Stimulation (TMS)
- SPRY TMS
- TMS Neuronavigation
- MEGA-TMS
- Neuro-MEP Software
- TMS compatible EEG
- MEGA-EMG
- MagXite
- Vital Sign Monitoring
- HealthDot
- Mobile EEG
- MOBILE EEG Overview
- SMARTING PRO Line
- Smarting S
- Smarting Spectra
- Functional Near Infrared Spectroscopy
- NIRSIT System
- NIRSIT LITE
- NIRSIT ON
- Artinis Systems
- Soterix Medical Workshop
- Soterix Medical Workshop NYC 2026
- Webinars
- Unique tDCS by Soterix Medical
- Home-tDCS Depression Trial
- Soterix Medical Virtual Booth
- Transcutaneous Auricular Vagus Nerve Stimulation (taVNS)
- Pre-Clinical
- Animal DCS Overview
- Animal DCS / tES
- Animal Accessories
- Animal Accessories
- Animal Transcranial Magnetic Stimulation System
- Animal tFUS
- Animal Transcranial Focused Ultrasound Stimulation (tFUS) system
- RK-20 Animal tFUS
- Linear Current Isolator
- EDUCATION TECHNOLOGY
- Smart Mobility
- Support
- Contact us
- Distributors
- Cart
- Manuals
- Legacy
- Legacy Devices
- Research Video Library
- Configure Products
- Request Product Information
- Device registration
- News Room
- Press
- Events
- Newsletter
- Clinical trials
- Publications
- Webinars
- Images
- About us
- EASYpad™
- EASYstrap
- Elastic Fastener Set
- SNAPpad™
- SNAPstrap
- Carbon Rubber Electrode
- EASYkit
- EASYcase
- Clinical Neuromodulation CART
- Breakout Box
- Banana to Din Adapter
- SMI Signal Isolator
- SNAPheadset
- HD-Electrode
- HD1 Electrode Holder
- HD1-Biosemi Electrode Holder
- HD-Cap
- HDM Electrode Holder
- HDM-Biosemi Electrode Holder
- HD-GEL
- 4x1 Input Cable
- MxN-33 Output Cable
- HD-tES Soft Base MRI Electrode Holder
- HD-tES MRI Electrode Holders
- MxN-33 Cable Disconnector
- SMI Chronos Adapter
- GVS HEADstrap
- GVS Headset
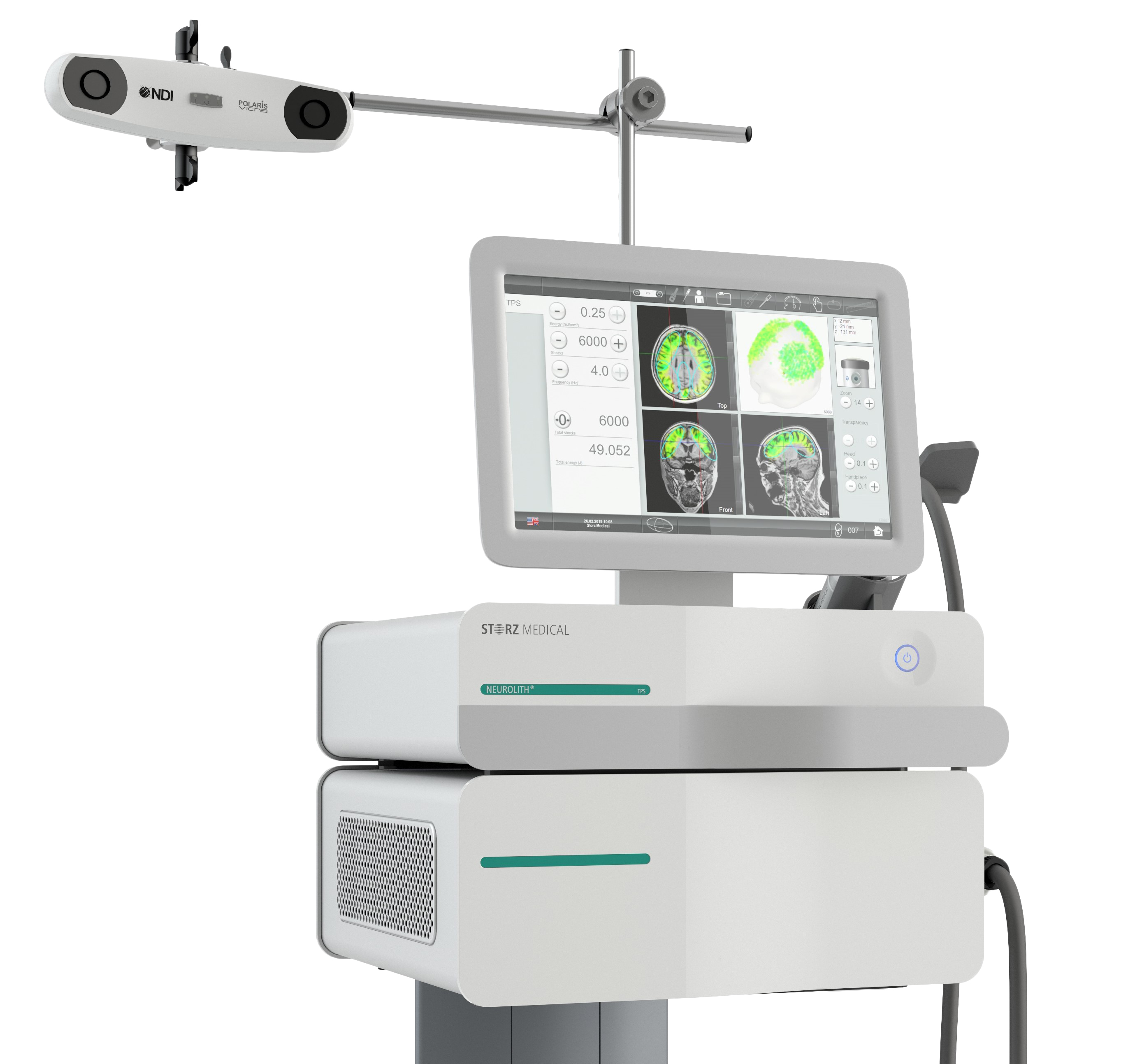
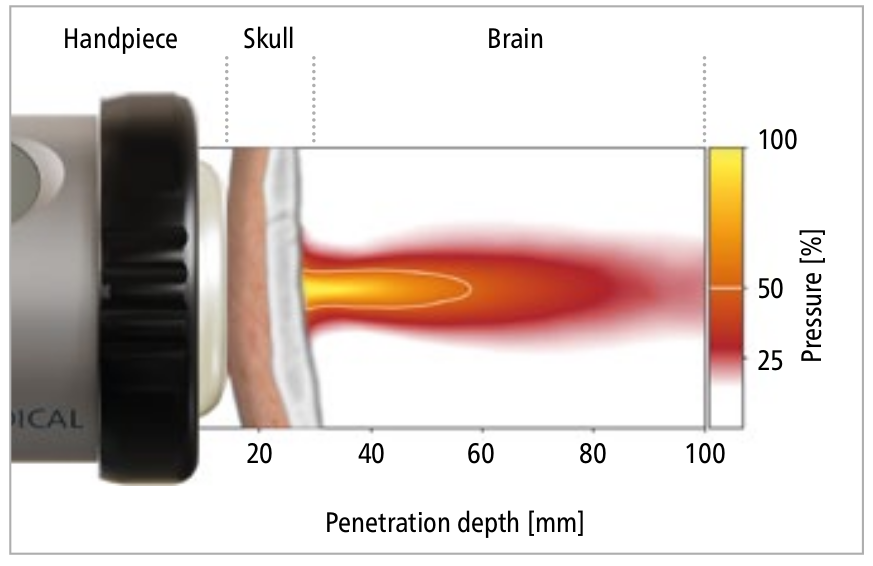
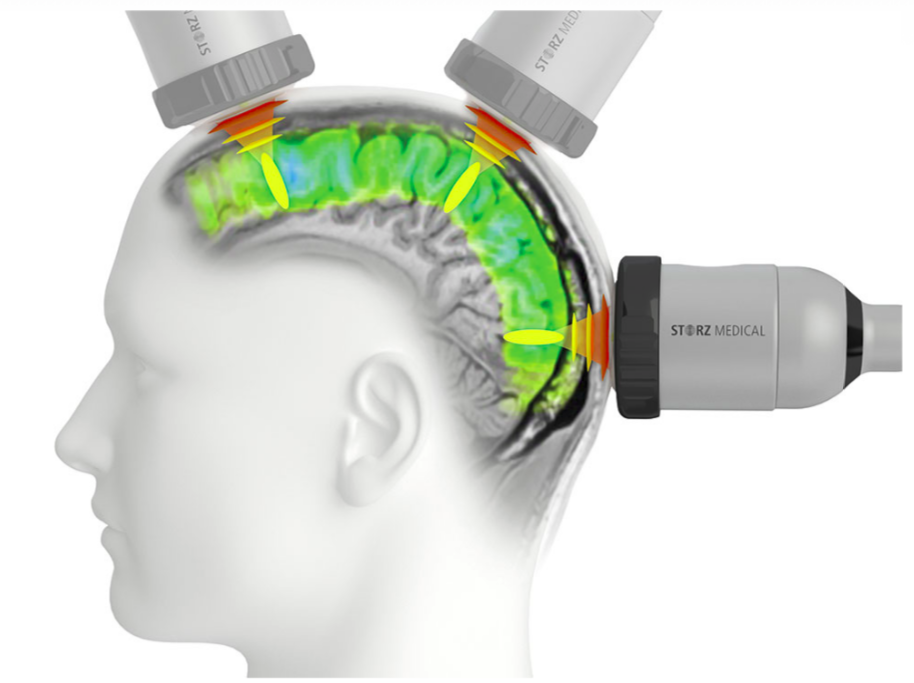
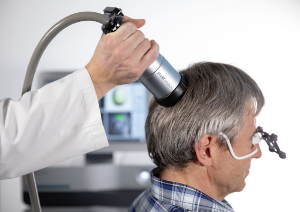
Research Transcranial Pulse Stimulation
Transcranial Pulse Stimulation
 Transcranial Pulse Stimulation
Transcranial Pulse Stimulation