Worldwide
LoginNon-invasive TMS Motor Mapping from Soterix Medical empowers clinicians to determine motor functions accurately on an individual basis and without line-of-sight occlusion.

Robust Functional Localization
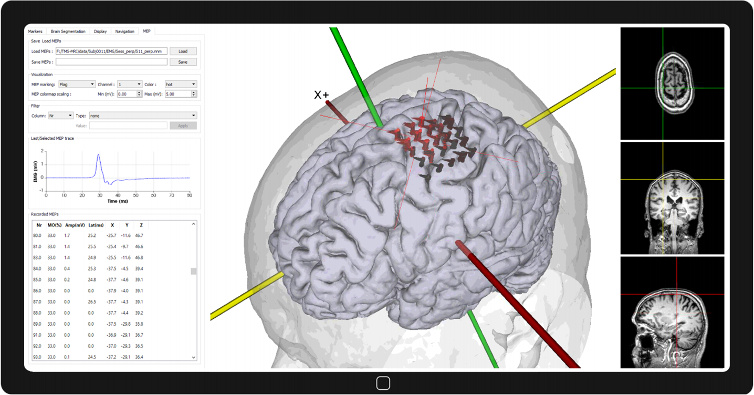
Use a grid placed over the motor region to simply move the TMS coil from one grid location to another. In addition to TMS pulse location and coil direction, additional information such as motor output percentage, amplitude, latency, etc. are saved for further analysis.
Explore how TMS Motor Mapping Works
Single pulse TMS combined with neuronavigation allows to ‘mapping’, i.e. to locate and outline, functional areas of the cortex. This feature is used for applications ranging from basic neurophysiology / plasticity assessment, brain surgery planning, or to understand motor recovery after stroke. It is known that cortical locations corresponding to neurologic functions can vary significantly among individuals. In addition, lesions and tumors may distort underlying anatomical features, thereby further hindering localization of key functions. TMS Motor Mapping therefore presents a rational way to assess and locate the exact cortical area responsible for a specific function. Motor mapping with TMS begins with acquiring high-resolution structural MRI data sufficient to provide necessary anatomical detail. The MRI is imported into the Neural Navigator system and a 3D model of the patient showing the skin is constructed immediately. A patient-specific file is created by marking facial landmarks on the scalp like the tip of the nose, tragus of the ears, etc. and then using the in-built brain segmentation routine to visualize the brain. This file is created offline and prior to the procedure. In the patient room, surface electrodes are attached to the patient’s muscles for EMG recording. The exact location is determined by the muscles that the physician wants to study. This could range from the face, hand, arm, leg, and the foot muscles. To initiate navigated TMS, the patient-specific file created prior, is opened. The hand-held pointer is then moved close to the chosen facial landmarks on the patient’s skin one-by-one and the tip coordinates of the pointer captured. These tip coordinates are then aligned with the coordinates of the patients 3D model to begin navigation. Resting Motor Threshold (RMT) of the patient is then determined by delivering magnetic pulses from a TMS coil. It is typically defined as the minimum TMS stimulation intensity capable of generating an Motor Evoked Potential (MEP) in 50% of the cases. Motor mapping is then performed at 110% of the RMT. The location of the TMS pulses as well as the TMS coil direction are depicted as flags and additional information such as motor output percentage, amplitude, latency, etc. are saved. After motor mapping is complete, the physician processes the saved MEP data (for instance, less than or equal to a certain threshold, range, etc.) and determines the treatment option. The Neural Navigator is the only FDA cleared Neuronavigation System that is not affected by line-of-sight occlusion. The clearance was based on clinical evidence showing tracking and navigation system accuracy better than the predicate device. The Neural Navigator offers additional benefits such has highly intuitive software workflow, light weight, and having the smallest footprint in its class that allows fitting the system into tight spaces. The fully integrated solution from Soterix Medical combines Neural Navigator, MEGA-TMS (Single Pulse TMS device) with an EMG system. This combined offering reflects the only system that is built to accommodate the latest advance in TMS Motor Mapping.